Ring lithiation of 1,8-bis(dimethylamino)naphthalene: another side of the ‘proton sponge coin’†
Abstract
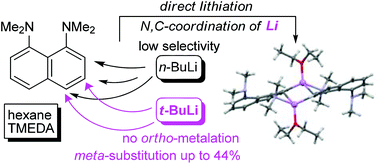
It has been found that 1,8-bis(dimethylamino)naphthalene (DMAN), unlike N,N-dimethylaniline, undergoes ring metallation in the n-BuLi–TMEDA–Et2O system with a low selectivity and in poor total yields. The situation is significantly improved in the t-BuLi–TMEDA–n-hexane system when 3- and 4-lithium derivatives become the only reaction products obtained in good yields. The formation of 3-Li-DMAN is especially desired since no method of direct meta-functionalization of DMAN is known to date. The relative stability and structure of DMAN lithium derivatives have been examined with the help of X-ray and multinuclear NMR measurements as well as DFT calculations.


 Please wait while we load your content...
Please wait while we load your content...