2,2′-Disilylazobenzenes featuring double intramolecular nitrogen⋯silicon coordination: a photoisomerizable fluorophore†
Abstract
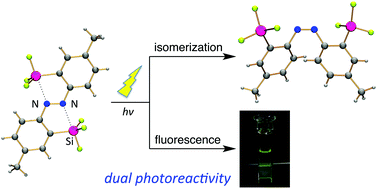
(E)-4,4′-Dimethyl-2,2′-disilylazobenzenes were synthesized. Double intramolecular N⋯Si coordination in the bis(fluorodimethylsilyl) and bis(trifluorosilyl) derivatives was confirmed using X-ray crystallographic analysis and 29Si NMR spectroscopy. In the absorption bands, due to the π,π* transitions, introduction of silyl groups was found to cause a bathochromic shift. In contrast to most azobenzenes, which do not fluoresce at all, the (E)-2,2′-bis(trifluorosilyl)azobenzene derivative with the N⋯Si coordination fluoresced a yellow-green colour at room temperature. Methyl and trifluorosilyl groups lowered the n and π* orbitals, as revealed by DFT calculations. As a result, the lowest singlet excitation energy state is found to be the allowed π,π* transition, different from the forbidden n,π* transition in general azobenzenes, as revealed by TD-DFT calculations. The allowed transition character of the lowest singlet excited state and moderately rigid conformation of the azo moiety, provided by the double N⋯Si coordination, account for the fluorescence emission. Nevertheless, the N⋯Si coordination is weak enough to be cleaved upon photoexcitation, and thus the (E)-2,2′-disilylazobenzenes undergo photoisomerization to the (Z)-isomers. Both the photoisomerization and fluorescence emission properties of the azobenzene moiety have been achieved for the first time. After photoisomerization of the (E)-2,2′-disilylazobenzenes to the corresponding (Z)-isomer, they do not fluoresce. This change in the fluorescence intensity upon photoisomerization is useful for the regulation of fluorescence properties. Therefore, this compound can be recognized as a unique photoisomerizable fluorophore to regulate the fluorescence intensity using a single light source.

 Please wait while we load your content...
Please wait while we load your content...