A high surface area flower-like Ni–Fe layered double hydroxide for electrocatalytic water oxidation reaction†
Abstract
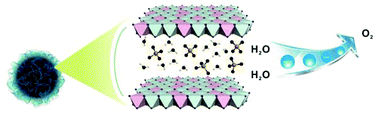
Layered double hydroxide has been used in a variety of areas, including but not limited to catalysis, energy storage, drug or gene delivery, water treatment, etc. Herein, we report a new simple hydrothermal method to prepare a high surface area flower-like Ni–Fe layered double hydroxide (LDH) assembled by nanosheets by using nickel alkoxide and FeSO4 as the only starting materials. It is free of alkaline solution and other additives for directing or supporting in the synthesis procedure. The formation mechanism of this flower-like LDH formed by ultrathin nanosheets is also discussed. Moreover, the as-obtained LDH material shows increased electrocatalytic activity and stability toward WOR in alkaline media compared with the materials prepared without a Ni alkoxide precursor or Fe precursor, namely α-Fe2O3 and Ni(OH)2, respectively. In addition, the electrocatalytic activity is demonstrated to be related to the molar ratio of Fe and Ni in the final Ni–Fe material, and the best activity is achieved when the ratio reaches 0.52 : 1. The phase compositions of the resulting Ni–Fex are discussed. Furthermore, the Ni–Fe LDH material reported herein might be employed as a promising noble-metal-free water oxidation catalyst to replace the IrOx material—the state-of-the-art water oxidation catalyst.

 Please wait while we load your content...
Please wait while we load your content...