Modeling of the hydrophobic microenvironment of water-soluble molybdoenzymes in an aqueous micellar solution†
Abstract
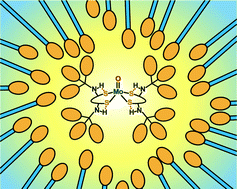
A toluene-soluble molybdenum(VI) complex containing a bulky hydrophobic substituent, (Et4N)2[MoVIO2{1,2-S2-3,6-(RCONH)2C6H2}2] (R = (4-tBuC6H4)3C), was dissolved in the hydrophobic core of a micelle in an aqueous medium and catalyzed the biomimetic reduction of an amine N-oxide by an NADH analog. The kinetic isotope effect of solvent water clearly indicates that water molecules are essential for catalysis and are involved in the rate-determining step.

 Please wait while we load your content...
Please wait while we load your content...