Six Zn(ii) and Cd(ii) coordination polymers assembled from a similar binuclear building unit: tunable structures and luminescence properties†
Abstract
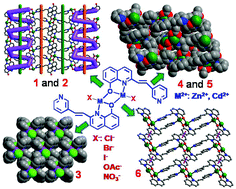
Six Zn(II) and Cd(II) coordination polymers were constructed by treating a 2-substituted 8-hydroxyquinolinate ligand containing a pyridyl group with zinc or cadmium salts, and characterized by a variety of techniques. Interestingly, based on a similar binuclear Zn(II) or Cd(II) building unit, the supramolecular structures of the six coordination polymers (1–6) exhibit an unprecedented structural diversification due to the different choices of metal salts. 1 and 2 represent a novel 2D framework containing 1D infinite right- and left-handed helical chains. 4 and 5 are 2D coordination frameworks based on binuclear Cd(II) building units. For 3 and 6, the L ligands can bridge binuclear building units forming a 1D infinite chain. Interestingly, the adjacent Cd2O2 planes of the 1D chain in 6 are in parallel with each other, while the dihedral angle between the two Zn2O2 planes in 3 is 83.43°. Photoluminescence properties revealed that the six coordination polymers exhibit redshifted emission maximum compared with the free ligand HL, which can be ascribed to an increased conformational rigidity and the fabrication of coplanar binuclear building units M2L2 in 1–6. Coordination polymers 1–6 also display distinct fluorescence lifetimes and quantum yields because of their different metal centers and supramolecular structures.

 Please wait while we load your content...
Please wait while we load your content...