A perfectly aligned 63 helical tubular cuprous bromide single crystal for selective photo-catalysis, luminescence and sensing of nitro-explosives†
Abstract
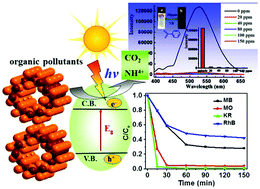
A perfectly aligned 63 helical tubular cuprous bromide single crystal has been synthesized and characterized, which can selectively decompose negatively charged dyes of Methyl Orange (MO) and Kermes Red (KR), and the photocatalytic efficiency is higher than that of nanosized (∼25 nm) TiO2 and ZnO. The direction and magnitude of the dipole moments as well as the band structure were calculated to reveal high photocatalytic efficiency. Moreover, luminescence studies indicate that the CuBr tube materials show very strong yellowish green emissions in the solid state and emulsion even at room temperature, and exhibit extremely high detection sensitivity towards nitro-explosives via fluorescence quenching. Detectable luminescence responses were observed at a very low concentration of 20 ppm with a high quenching efficiency of 94.90%. The results suggest that they may be promising multifunctional materials for photo-catalysis, luminescence and sensing of nitro-explosives.

 Please wait while we load your content...
Please wait while we load your content...