Phosphine complexes of lone pair bearing Lewis acceptors
Abstract
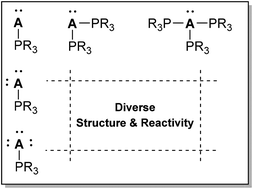
An overview of the synthesis, structures and reaction chemistry of coordination complexes featuring an acceptor with at least one lone pair and at least one phosphine donor is presented. One or more examples of complexes have been structurally-characterized for the majority of p-block elements but few are known for most elements. The unusual condition of a p-block element centre accommodating a lone pair of electrons and offering a low energy LUMO gives the element centre the potential to behave as both a Lewis acid and a Lewis Base. The structural diversity and reactivity of the phosphine complexes highlights new directions in main group chemistry and by comparison with transition metal coordination chemistry, the featured complexes demonstrate significant configurational and stereochemical flexibility. Ligand exchange, oxidation and reduction chemistry at the lone pair bearing acceptor centre reveals unusual reactivity and an interesting class of ligands and inorganic reagents, with new possibilities for catalysis or small molecule activation.

 Please wait while we load your content...
Please wait while we load your content...