Photocatalytic hydrogen evolution from a cobalt/nickel complex with dithiolene ligands under irradiation with visible light†
Abstract
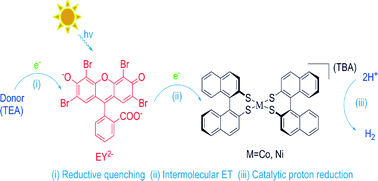
Two novel metal dithiolene complexes, namely NtBu4[M(BNT)2] [M = Co/Ni, (BNT = (R)-1, 1′-binaphthalene-2,2′-dithiol)], have been synthesized and characterized. An efficient homogeneous photocatalytic system was constructed by a combination of the noble-metal-free target complexes as water reduction catalysts, xanthene dyes as the photosensitizer and triethylamine (TEA) as the sacrificial electron donor under irradiation with visible light (λ > 420 nm). Maximum H2 evolution of 495 and 676 turnovers (vs. catalyst) were recorded for each catalyst, respectively, under optimal conditions in CH3CN/H2O (1 : 1, v/v) after 4 h of irradiation. Furthermore, the mechanism of the H2 evolution was also briefly discussed alongside fluorescence spectra and cyclic voltammetry studies.

 Please wait while we load your content...
Please wait while we load your content...