High rectification in organic diodes based on liquid crystalline phthalocyanines†
Abstract
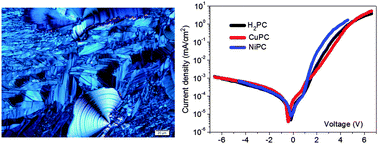
The optical and electrical properties of mesogenic metal-free and metalated phthalocyanines (PCs) with a moderately sized and regioregular alkyl periphery were investigated. In solution, the individualized molecules show fluorescence lifetimes of 4–6 ns in THF. When deposited as solid thin films the materials exhibit significantly shorter fluorescence lifetimes with bi-exponential decay (1.4–1.8 ns; 0.2–0.4 ns) that testify to the formation of aggregates via π–π intermolecular interactions. In diode structures, their pronounced columnar order outbalances the unfavorable planar alignment and leads to excellent rectification behavior. Field-dependent charge carrier mobilities are obtained from the J–V curves in the trap-limited space-charge-limited current regime and demonstrate that the metalated PCs display an improved electrical response with respect to the metal-free homologue. The excited-state lifetime characterization suggest that the π–π intermolecular interactions are stronger for the metal-free PC, confirming that the metallic centre plays an important role in the charge transport inside these materials.

 Please wait while we load your content...
Please wait while we load your content...