Disulfuric acid dissociated by two water molecules: ab initio and density functional theory calculations†
Abstract
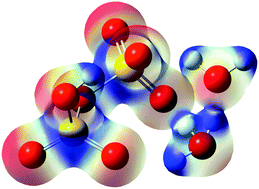
We have studied geometries, energies and vibrational spectra of disulfuric acid (H2S2O7) and its anion (HS2O7−) hydrated by a few water molecules, using density functional theory (M062X) and ab initio theory (SCS-MP2 and CCSD(T)). The most noteworthy result is found in H2S2O7(H2O)2 in which the lowest energy conformer shows deprotonated H2S2O7. Thus, H2S2O7 requires only two water molecules, the fewest number of water molecules for deprotonation among various hydrated monomeric acids reported so far. Even the second deprotonation of the first deprotonated species HS2O7− needs only four water molecules. The deprotonation is supported by vibration spectra, in which acid O–H stretching peaks disappear and specific three O–H stretching peaks for H3O+ (eigen structure) appear. We have also kept track of variations in several geometrical parameters, atomic charges, and hybrid orbital characters upon addition of water. As the number of water molecules added increases, the S–O bond weakens in the case of H2S2O7, but strengthens in the case of HS2O7−. It implies that the decomposition leading to H2SO4 and SO3 hardly occurs prior to the 2nd deprotonation at low temperatures.

 Please wait while we load your content...
Please wait while we load your content...