Energy dissipative photoprotective mechanism of carotenoid spheroidene from the photoreaction center of purple bacteria Rhodobacter sphaeroides†
Abstract
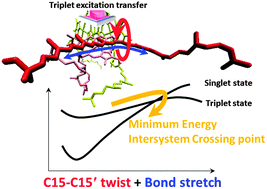
Carotenoid spheroidene (SPO) functions for photoprotection in the photosynthetic reaction centers (RCs) and effectively dissipates its triplet excitation energy. Sensitized cis-to-trans isomerization was proposed as a possible mechanism for a singlet–triplet energy crossing for the 15,15′-cis-SPO; however, it has been questioned recently. To understand the dissipative photoprotective mechanism of this important SPO and to overcome the existing controversies on this issue, we carried out a theoretical investigation using density functional theory on the possible triplet energy relaxation mechanism through the cis-to-trans isomerization. Together with the earlier experimental observations, the possible mechanism was discussed for the triplet energy relaxation of the 15,15′-cis-SPO. The result shows that complete cis-to-trans isomerization is not necessary. Twisting the C15–C15′ bond leads to singlet–triplet energy crossing at ϕ(14,15,15′,14′) = 77° with an energy 32.5 kJ mol−1 (7.7 kcal mol−1) higher than that of the T1 15,15′-cis minimum. Further exploration of the minimum-energy intersystem crossing (MEISC) point shows that triplet relaxation could occur at a less distorted structure (ϕ = 58.4°) with the energy height of 26.5 KJ mol−1 (6.3 kcal mol−1). Another important reaction coordinate to reach the MEISC point is the bond-length alternation. The model truncation effect, solvent effect, and spin–orbit coupling were also investigated. The singlet–triplet crossing was also investigated for the 13,14-cis stereoisomer and locked-13,14-cis-SPO. We also discussed the origin of the natural selection of the cis over trans isomer in the RC.

 Please wait while we load your content...
Please wait while we load your content...