Acid-catalyzed carboxylic acid esterification and ester hydrolysis mechanism: acylium ion as a sharing active intermediate via a spontaneous trimolecular reaction based on density functional theory calculation and supported by electrospray ionization-mass spectrometry†
Abstract
By DFT calculation, we found that acid-catalyzed carboxylic acid esterification and ester hydrolysis are brief two-step reactions. First, the carboxylic acid hydroxyl-oxygen or ester alkyl-oxygen is protonated, which generates a highly active acylium ion. The protonation requires an activation energy (Ea) of 4–10 kcal mol−1, and is the rate-controlling step of the esterification or hydrolysis. Sequentially, the acylium ion spontaneously reacts with two alcohol or two water molecules to form a neutral product molecule; this is a trimolecular reaction. The acylium ion is the highly active intermediate shared by esterification and hydrolysis. ESI-MS data for several typical carboxylic acids confirmed that their acylium ions are easily generated. For 2,4,6-trialkylbenzoic acid and its ester, the two unsubstituted carbons in the benzene ring are very easily protonated, and we have thus revealed the root of the success of Newman's method. Based on these results, the popular esterification and hydrolysis mechanism in organic chemistry textbooks is incorrect.
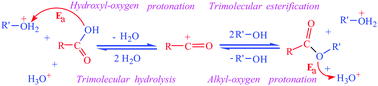

 Please wait while we load your content...
Please wait while we load your content...