A study by electrical conductivity measurements of the semiconductive and redox properties of Nb-doped NiO catalysts in correlation with the oxidative dehydrogenation of ethane†
Abstract
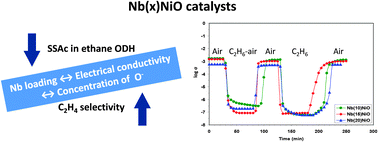
Nb-doped nickel oxides with Nb contents in the range from 1 to 20% and, for comparison, pure NiO, were characterized using in situ electrical conductivity measurements in correlation with their catalytic performances for the oxidative dehydrogenation (ODH) of ethane into ethylene. Their electrical conductivity was studied as a function of temperature and oxygen partial pressure and was followed with time during sequential exposures to air, ethane–air mixture (reaction mixture) and pure ethane in conditions similar to those of catalysis. All the oxides were p-type semiconductors under air. Their electrical conductivity in the reaction temperature range decreased in the following order: NiO > Nb(1)NiO > Nb(5)NiO > Nb(10)NiO > Nb(15)NiO > Nb(20)NiO. This correlates well with the catalytic activity expressed as the intrinsic rate of ethane consumption. All the catalysts were partially reduced under the reaction mixture in the reaction temperature range, an inverse correlation between their conductivity in these conditions and the ODH selectivity being observed. The ODH reaction of ethane takes place via a heterogeneous redox mechanism involving the surface lattice O− species.

 Please wait while we load your content...
Please wait while we load your content...