The role of tryptophans in the UV-B absorption of a UVR8 photoreceptor – a computational study†
Abstract
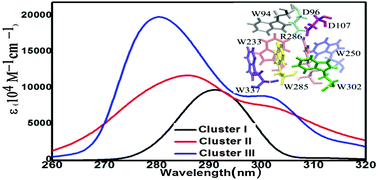
Arabidopsis thaliana UV RESISTANCE LOCUS8 (UVR8) has been identified as a photoreceptor for ultraviolet-B (UV-B). Tryptophan (Trp) residues have been shown to play a critical role in the response to UV-B irradiation in UVR8. In this work, we explore the spectroscopic behaviors of Trps in different protein environments of the UVR8 structure using the time-dependent density functional tight-binding (TD-DFTB) scheme. We show that W233 exhibits the longest absorption wavelength, highlighting its potential as a terminal Trp chromophore in the UV-B harvesting antenna. Our electronic and optical property analyses using various amino acid models support the important roles of W285 and W233 in sensing UV-B light at longer absorption wavelengths (∼290 nm). We also provide evidence for the specific function of W94 in absorption at the longest wavelengths (305.8 nm in cluster II and 304.5 nm in cluster III). To these findings, we also add information about the influence of the arginine and aspartic acid residues surrounding the Trp pyramid on the particular absorption bands (280–300 nm) that are characteristic of the UV-B photoreceptor.


 Please wait while we load your content...
Please wait while we load your content...