Molecular thermodynamics of metabolism: quantum thermochemical calculations for key metabolites†
Abstract
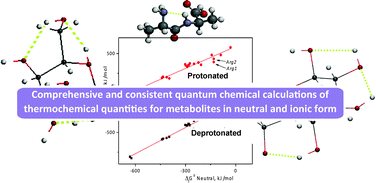
The present work is the first of a series of papers aiming at a coherent and unified development of the thermodynamics of metabolism and the rationalization of feasibility analysis of metabolic pathways. The focus in this part is on high-level quantum chemical calculations of the thermochemical quantities of relatively heavy metabolites such as amino acids/oligopeptides, nucleosides, saccharides and their derivatives in the ideal gas state. The results of this study will be combined with the corresponding hydration/solvation results in subsequent parts of this work in order to derive the desired thermochemical quantities in aqueous solutions. The above metabolites exist in a vast conformational/isomerization space including rotational conformers, tautomers or anomers exhibiting often multiple or cooperative intramolecular hydrogen bonding. We examine the challenges posed by these features for the reliable estimation of thermochemical quantities. We discuss conformer search, conformer distribution and averaging processes. We further consider neutral metabolites as well as protonated and deprotonated metabolites. In addition to the traditional presentation of gas-phase acidities, basicities and proton affinities, we also examine heats and free energies of ionic species. We obtain simple linear relations between the thermochemical quantities of ions and the formation quantities of their neutral counterparts. Furthermore, we compare our calculations with reliable experimental measurements and predictive calculations from the literature, when available. Finally, we discuss the next steps and perspectives for this work.

 Please wait while we load your content...
Please wait while we load your content...