Chemisorptive enantioselectivity of chiral epoxides on tartaric-acid modified Pd(111): three-point bonding
Abstract
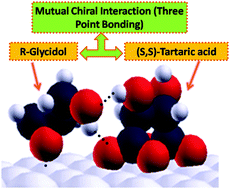
The chemisorption of two chiral molecules, propylene oxide and glycidol, is studied on tartaric-acid modified Pd(111) surfaces by using temperature-programmed desorption to measure adsorbate coverage. It is found that R-glycidol shows preferential enantioselective chemisorption on (S,S)-tartaric acid modified Pd(111) surfaces, while propylene oxide does not adsorb enantioselectively. The enantioselectivity of glycidol depends on the tartaric acid coverage, and is exhibited for low tartaric acid coverages indicating that the bitartrate phase is responsible for the chiral recognition. The lack of enantioselectivity when using propylene oxide as a chiral probe implies that the enantiospecific interaction between glycidol and bitartate species is due to hydrogen-bonding interactions of the –OH group of glycidol. Scanning tunneling microscopy images were collected for tartaric acid adsorbed on Pd(111) under the same experimental conditions as used for enantioselective experiments. When tartaric acid is dosed at room temperature and immediately cooled to 100 K for imaging, individual bitartrate molecules were found. Density functional theory (DFT) calculations show that bitartrate binds to Pd(111) through its carboxylate groups and the –OH groups are oriented along the long axis of the bitartrate molecule. An enantiospecific interaction is found between glycidol and bitartate species where R-glycidol binds more strongly than S-glycidol to (S,S)-bitartate species by simultaneously forming hydrogen bonds with both the hydroxyl and carboxylate groups, thereby providing three-point bonding.

 Please wait while we load your content...
Please wait while we load your content...