Proton irradiation of DNA nucleosides in the gas phase†
Abstract
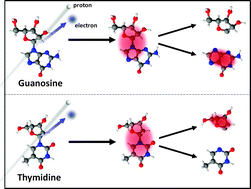
The four DNA nucleosides guanosine, adenosine, cytidine and thymidine have been produced in the gas phase by a laser thermal desorption source, and irradiated by a beam of protons with 5 keV kinetic energy. The molecular ions as well as energetic neutrals formed have been analyzed by mass spectrometry in order to shed light on the ionization and fragmentation processes triggered by proton collision. A range of 8–20 eV has been estimated for the binding energy of the electron captured by the proton. Glycosidic bond cleavage between the base and sugar has been observed with a high probability for all nucleosides, resulting in predominantly intact base ions for guanosine, adenosine, and cytidine but not for thymidine where intact sugar ions are dominant. This behavior is influenced by the ionization energies of the nucleobases (G < A < C < T), which seems to determine the localization of the charge following the initial ionization. This charge transfer process can also be inferred from the production of protonated base ions, which have a similar dependence on the base ionization potential, although the base proton affinity might also play a role. Other dissociation pathways have also been identified, including further fragmentation of the base and sugar moieties for thymidine and guanosine, respectively, and partial breakup of the sugar ring without glycosidic bond cleavage mainly for adenosine and cytidine. These results show that charge localization following ionization by proton irradiation is important in determining dissociation channels of isolated nucleosides, which could in turn influence direct radiation damage in DNA.

 Please wait while we load your content...
Please wait while we load your content...