A single-crystal-to-single-crystal Diels–Alder reaction with mixed topochemical and topotactic behaviour†
Abstract
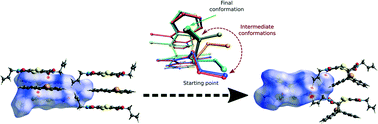
Electron donor/acceptor (EDA) interactions have been found to be very useful in engineering reactive heteromolecular crystals, but few examples have been reported in the literature. By utilising EDA interactions, crystals of charge-transfer (CT) complexes were formed with bis(N-allylimino)-1,4-dithiin as the electron acceptor and 9-bromoanthracene as the electron donor. The CT complex crystallised in the monoclinic P21/n space group with the crystal structure consisting of stacks of alternating electron donor and acceptor molecules in a 1 : 1 ratio. These crystals are able to undergo a solid-state Diels–Alder reaction with bis(N-allylimino)-1,4-dithiin as the dienophile and 9-bromoanthracene as the diene. Examination of close contacts indicates that the diene can theoretically react with the dienophile above or below it within a stack as the reaction distances are less than 3.5 Å in both directions. A single crystal was selected and allowed to react at 30 °C, was analysed at various states of conversion by single-crystal X-ray diffraction, and was found to react by approximately 10% every 6 days, with the reaction occurring in a single direction along the CT stack axis. The solid-state reaction creates a void space which leads to a molecular conformational change within the crystal. Consequently, the single crystal started to show significant signs of deterioration after approximately 28% conversion but remained intact upon further reaction and was found to anneal as 100% conversion was approached, leading to the formation of new intermolecular interactions not present in the starting crystal. The solid-state reaction occurs topochemically when fewer than 28% or more than 80% of the molecules have reacted, with minimal motion during the reaction. In the conversion range of 28–80%, the reaction occurs in an almost topotactic manner with significant molecular motion and associated crystal deterioration.
- This article is part of the themed collection: Single-Crystal-to-Single-Crystal Transformations

 Please wait while we load your content...
Please wait while we load your content...