The role of surface chemistry in crystal morphology and its associated properties†
Abstract
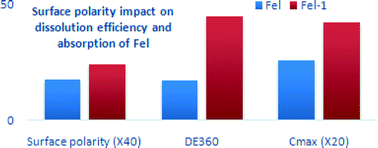
Acetonitrile induced a remarkable enhancement of the {11−1} crystal facet of felodipine. This facet was polar in nature as concluded from surface chemistry determination and resulted in significant improvement in the dissolution rate and oral bioavailability (p < 0.05). This conclusion was supported with surface chemistry determination by X-ray photoelectron spectroscopy and Hirshfeld surface analysis.

 Please wait while we load your content...
Please wait while we load your content...