Selective growth of TiO2 beads on Ag nanowires and their photocatalytic performance†
Abstract
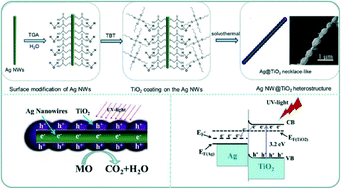
In this paper, TiO2 beads were deposited on Ag nanowires to create necklace-like Ag nanowire@TiO2 (Ag NW@TiO2) heterostructures for the first time. They were fabricated via a two-step synthetic method, including the preparation of uniform Ag NWs and the deposition of TiO2 on the Ag surface. Thioglycolic acid (TGA) molecules were used as binding agents to solve the tricky issue of substantial lattice mismatch between the Ag NW core and the TiO2 shell. The amount of water and the permittivity of the solvents are found to be important factors for the formation of the necklace-like heterostructures. Moreover, by varying the amount of titanium butoxide (TBT), the diameter of the TiO2 beads can be feasibly changed. The photocatalytic performance of necklace-like Ag NW@TiO2 was evaluated via degradation of methyl orange (MO) under ultraviolet (UV) light. The results reveal that the molar ratio of TiO2 to Ag contributes to the synergistic effect on the photocatalytic activity, and the best Ti/Ag ratio is found to be 2.8 for the highest photocatalytic performance of the Ag NW@TiO2 heterostructures.

 Please wait while we load your content...
Please wait while we load your content...