On molecular complexes derived from amino acids and nicotinamides in combination with boronic acids†‡
Abstract
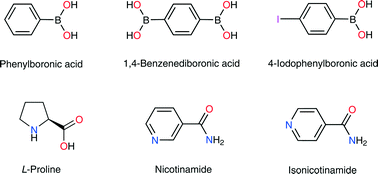
Cocrystallization experiments of three representative aromatic boronic acids, namely phenylboronic acid (PBA), 1,4-benzenediboronic acid (BDBA) and 4-iodophenylboronic acid (IPBA), with a series of essential amino acids, nicotinamide (NA) and isonicotinamide (INA) gave a total of nine molecular complexes of the compositions: PBA–PRO (1 : 1), PBA–PRO–H2O (1 : 1 : 1), α-BDBA–PRO (1 : 2), β-BDBA–PRO (1 : 2), α-IPBA–PRO (1 : 1), β-IPBA–PRO (1 : 1), BDBA–INA (1 : 2), IPBA–INA (1 : 1) and IPBA–NA (1 : 1), where PRO = L-proline. Of these, α-BDBA–PRO/β-BDBA–PRO and α-IPBA–PRO/β-IPBA–PRO were true polymorphs. Experiments varying the solvent in the screening process showed that the formation of the polymorphs is influenced strongly by the presence/absence of water. For the cocrystalline phases with L-proline, the structural characterization revealed that the molecular components were connected into high-dimensional hydrogen bonded networks. As expected, the charge-assisted –B(OH)2⋯carboxylate heterosynthon is dominant and was found to display varying degrees of distortion from planarity. The α-IPBA–PRO/β-IPBA–PRO polymorphs displayed a similar crystal packing, with differences only in one direction of the crystal lattice (AAAA versus ABAB stacking). The cocrystals with nicotinamide and isonicotinamide exhibited 1D or 2D hydrogen bonded layers, which were formed through (B)O–H⋯pyridine, B(OH)2⋯amide, amide⋯amide and/or single-bridged (B)O–H⋯O(H)B interactions. Comparison between the crystal structures of IPBA–INA and IPBA–NA revealed that the position of the heteroatom in the (iso)nicotinamide not only controlled the final crystalline form but also produced different hydrogen bonding interactions. In vitro toxicology studies showed that the cell viability is conserved when human kidney or hepatic cells are treated even with high concentrations of the boronic acids examined herein. However, some boronic acids exhibited teratogenicity in chicken embryos.
- This article is part of the themed collection: Polymorphism

 Please wait while we load your content...
Please wait while we load your content...