Facile synthesis of a genuinely alkane-soluble but isolable lithium hydride transfer reagent†
Abstract
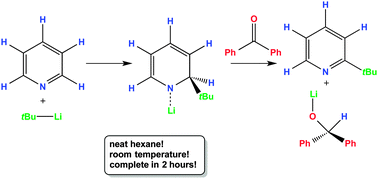
1-Lithio-2-butyl-1,2-dihydropyridines, typically formed as intermediates in the nucleophilic substitution (addition/elimination) of pyridine with (n- or t-)butyl lithium, have been isolated and comprehensively characterized. The linear substituted isomer is polymeric while the branched substituted isomer is a cyclotrimer. The lower oligomerization of the latter complex confers exceptional hexane solubility making it an excellent lithium hydride source in non-polar, aliphatic media. A Me6TREN stabilized monomer of the t-butyl complex represents the first 1,2-dihydropyridyllithium complex to be characterized crystallographically.
- This article is part of the themed collection: 2015 Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...