One-step electrochemical preparation of a reduced graphene oxide/poly(sulfosalicylic acid) nanocomposite film for detection of acetaminophen and its application in human urine and serum studies†
Abstract
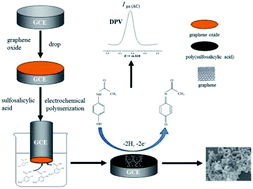
A mild preparation tactic was developed for the fabrication of a reduced graphene oxide (rGO) and poly(sulfosalicylic acid) (PSA) nanocomposite film by a one-step electrochemical method. The nanocomposite film was characterized by scanning electron microscopy (SEM), Fourier transform infrared (FTIR) spectroscopy, and electrochemical methods. The electrochemical properties of the nanocomposite were evaluated by means of cyclic voltammetry (CV) and differential pulse voltammetry (DPV). Based on the synergistic effect of the rGO and PSA nanocomposite film, a sensitive electrochemical sensor for acetaminophen (AC) was successfully fabricated. The electrochemical reaction of AC at a glassy carbon electrode (GCE) modified with the PSA and rGO nanocomposite film (PSA/rGO/GCE) was proved to be a surface-controlled process involving the same number of protons and electrons. Under optimum experimental conditions, the anodic peak currents were linear over the AC concentrations ranging from 0.5 to 300 μM, with a limit of detection (LOD) of 0.041 μM (S/N = 3). Furthermore, the modified electrode was demonstrated to be feasible for analytical purposes in real samples. A linear calibration curve was obtained for the determination of AC in urine within the range of 1 to 300 μM with a LOD of 0.36 μM under optimized conditions. In addition, the PSA/rGO/GCE was demonstrated in human serum with satisfactory results.

 Please wait while we load your content...
Please wait while we load your content...