1,8-Naphthyridinic fluorescent ‘turn-on’ and ‘turn-off’ chemosensors for detection of F− and Hg2+ ions mimicking INHIBIT molecular logic behaviour†
Abstract
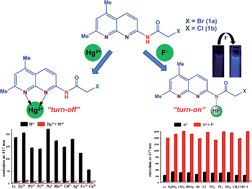
Two new receptors viz. 2-bromoacetamido-5,7-dimethyl-1,8-naphthyridine (1a) and 2-chloroacetamido-5,7-dimethyl-1,8-naphthyridine (1b) containing 1,8-naphthyridine as fluorophore unit have been designed and synthesised. These receptors exhibited very high selectivity for F− ions among the various anions tested such as H2PO4−, OAc−, Cl−, Br−, I−, HSO4−, PF6−, ClO4−, BF4− and NO3− owing to anion-induced deprotonation of the amide NH by F− ions. Among the various cations tested (Na+, Ag+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+, Hg2+, Pb2+ and Mn3+), 1a exhibited selective detection of Hg2+ ions by binding with N1 and N8 atoms of the naphthyridine moiety. Receptor 1a has been explored as a dual-ion detector for F− and Hg2+ ions in two contrasting modes (fluorescence ‘turn-on’ and ‘turn-off’) which is supported by spectroscopic and lifetime measurements. 1a exhibited lower detection limits for F− (0.4 ppm) and Hg2+ (2.4 ppm) ions. These receptors were recovered from anionic species of 1a formed after addition of F− ions by treating with protic solvents such as CH3OH. The mode of binding was also confirmed by spectroscopic and DFT studies. The chemical inputs of F− and Hg2+ ions generate an output which satisfies the condition of two-input INHIBIT logic operators.

 Please wait while we load your content...
Please wait while we load your content...