Detection of PETN and RDX using a FRET-based fluorescence sensor system†
Abstract
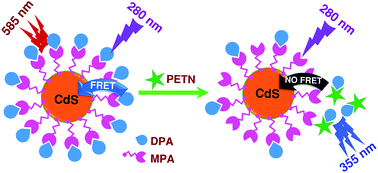
Most of the fluorescence based detection of explosives involves the detection of nitro-aromatic compounds, such as trinitrotoluene (TNT) and dinitrotoluene (DNT). Here, we report a Förster resonance energy transfer (FRET)-based nanosensor system for the highly selective detection of powerful explosives, such as PETN (pentaerythritol tetranitrate) and RDX (cyclotrimethylenetrinitramine). The nanosensor system is composed of cadmium sulfide quantum dots (CdS QDs) and diphenylamine (DPA). Initially, the inherent fluorescence of DPA was quenched by resonance energy transfer to the CdS QDs. During detection, due to the strong interaction of DPA with PETN/RDX, the FRET was turned-off and was accompanied by the recovery of the donor's (DPA) fluorescence. This provides an opportunity to follow the detection in a two-way manner, either the decrease in the FRET intensity at ∼585 nm or the evolution of fluorescence at ∼355 nm. The detection limits for PETN and RDX were found to be 10 nM and 20 nM, respectively. The fluorescence lifetime measurements confirmed that the energy transfer process is effective in the CdS QD–DPA sensor system. The details of the molecular interactions, between QD–DPA and DPA–analytes, were established using infrared spectroscopy. The easy one pot synthesis method of CdS QDs, excellent selectivity and very good sensitivity make the present sensor system attractive.

 Please wait while we load your content...
Please wait while we load your content...