Synthesis of model humic substances: a mechanistic study using controllable H/D exchange and Fourier transform ion cyclotron resonance mass spectrometry†
Abstract
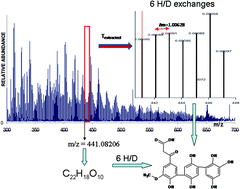
The products of the oxidative coupling of phenols are frequently used as synthetic analogues to natural humic substances (HS) for biomedical research. However, their molecular compositions and exact structures remain largely unknown. The objective of this study was to develop a novel approach for the molecular-level analysis of phenolic polymerisates that is capable of inventorying molecular constituents and resolving their distinct structural formulas. For this purpose, we have synthesized the model HS using the oxidative coupling of a specifically designed phenylpropanoic monomer, 3-(4-hydroxy-3-methoxyphenyl)-3-oxopropionic acid, to hydroquinone. We have characterized the synthesized model HS using high resolution Fourier transform ion cyclotron resonance mass spectrometry (FTICR MS), 1H NMR spectroscopy, and controllable hydrogen/deuterium (H/D) exchange. We succeeded in the molecular inventory of the model HS. The assigned molecular formulas occupied the substantial space of CHO compositions in the Van Krevelen diagram with a maximum density found in the regions of tannins and lignins, resembling those of natural HS. To identify the exact structural formulas of the individual constituents in the model HS, we have applied selective H/D exchange of non-labile backbone protons by a choice of basic or acidic catalytic conditions followed by FTICR MS. The determined formulas allowed us to verify the proposed pathways of hydroxylation and carboxylation in the course of the phenolic coupling and to identify the acetylation of aromatic rings as an important side reaction. We conclude that the proposed analytical approach may be used to identify the molecular carriers of biological activity within the phenolic polymerisates and eventually within natural HS.

 Please wait while we load your content...
Please wait while we load your content...