High pH sensing with water-soluble porpholactone derivatives and their incorporation into a Nafion® optode membrane†
Abstract
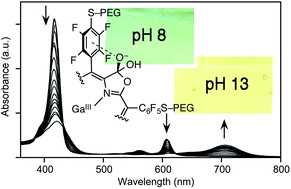
The known optical high pH sensing chromophores, free base and metal complexes (M = 2H, Zn(II), Pt(II)) of meso-tetrakis(pentafluorophenyl)porpholactone, and the as yet untested Ga(III) complex, were made freely water-soluble by derivatization at the aryl group with PEG chains. Their halochromic response profiles were determined and found to be surprisingly shifted toward greater base sensitivity when compared to the parent sensors in aqueous solution in the presence of a surfactant. Select PEG-derivatized chromophores were also incorporated into Nafion®-based membranes. The immobilized sensor was shown to be suitable for a moderately rapid (response time in minutes) sensing of high concentrations of hydroxides (pH 11 and above, up to 5 M NaOH concentrations). The lesser sensitivity of the indicators in the membrane is rationalized by the anionic nature of the membrane material. The membrane shows a perfectly reversible response and remains transparent and stable even under extended times of exposure to very caustic environments, and no leaching of the chromophore is observed. The membrane might find use in fiber optics-based optodes suitable for the monitoring of high hydroxide environments inside chemical reactors or fuel cells.

 Please wait while we load your content...
Please wait while we load your content...