Synthesis and characterization of mixed phase anatase TiO2 and sodium-doped TiO2(B) thin films by low pressure chemical vapour deposition (LPCVD)
Abstract
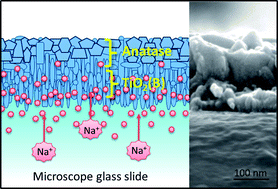
TiO2 thin films were synthesized using Low Pressure Chemical Vapour Deposition (LPCVD) onto glass substrates. Titanium isopropoxide (TTIP) and N2 gas were used as the precursor and carrier gas respectively. The effects of reaction temperature, carrier gas flow rate and deposited area were studied. SEM, TEM, powder XRD and UV-Vis and Raman spectroscopy were employed to characterize the phase and morphology of the synthesized materials. The results show that a dual phase (sodium-doped TiO2(B) and anatase) nanocrystalline thin film was successfully prepared by LPCVD with needle- and polygonal plate-shape crystallites respectively. At the interface with the substrate, the thin film deposit exhibited a preferred orientation of TiO2(B) needles in the [001] direction with an average crystallite size of 50–80 nm in length and 5–10 nm in width, whilst the crystallite size of anatase polygonal-plates was around 200 nm. The optimal LPCVD condition for preparing this mixed phase of TiO2 was 550 °C (actual temperature) with a 1 mL s−1 N2 flow rate. A possible mechanism for the mixed-phase formation by LPCVD on the glass substrates is described as well as the implications for the production of self-cleaning structures.

 Please wait while we load your content...
Please wait while we load your content...