Fine-tuning the mechanofluorochromic properties of benzothiadiazole-cored cyano-substituted diphenylethene derivatives through D–A effect
Abstract
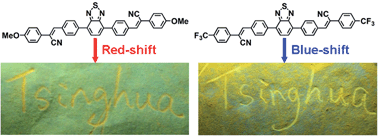
We synthesized three new benzothiadiazole-cored cyano-substituted diphenylethene derivatives (PT-OMe, PT-H, and PT-CF3) with different methoxy, hydrogen, and trifluoromethyl end groups, and the synthesis confirmed by standard spectroscopic methods. These end groups endowed them with different donor–acceptor (D–A) effects, and they provide them with a peculiar and completely opposite mechanofluorochromic property. Red-shifted mechanofluorochromic features were found in the PT-OMe and PT-H compounds, while on the contrary, PT-CF3 showed blue-shifted mechanofluorochromic behavior. The mechanofluorochromic mechanism was explored and attributed to the metastable state of the ground compounds and the crystalline-amorphous phase transformation between the original and ground states. Moreover, these derivatives showed reversible significant mechanofluorochromic properties and reproducibility between ground and annealed states, making them promising stimuli-responsive and smart luminescent materials for mechanosensors, fluorescence switches and light-emitting device applications. The introduction of the D–A effect strategy demonstrated in this work would provide a new path to fine tune the optical features of mechanofluorochromic materials with unique and diverse fluorescent properties.


 Please wait while we load your content...
Please wait while we load your content...