Design and mechanistic investigation of oxime-conjugated PAMAM dendrimers as the catalytic scavenger of reactive organophosphate†
Abstract
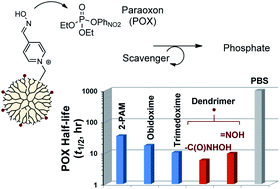
Pyridinium aldoxime (PAM) molecules constitute a group of small molecule antidotes essential for the treatment of reactive organophosphate (OP) poisoning. Their therapeutic efficacy stems from a combination of two activities: (i) reactivation of acetylcholine esterase inhibited by OP; (ii) scavenging of free OP. Here, we report the design, synthesis and in vitro functional characterization of fifth generation poly(amidoamine)dendrimer conjugates, each tethered with PAM or hydroxamate, as OP scavenging macromolecules. We chose paraoxon (POX) as the model OP, and performed extensive reaction kinetic studies in aqueous media to determine the activities and mechanisms of these dendrimer conjugates in POX hydrolysis with 1H NMR, UV-vis and LCMS/MS methods. Quantitative kinetic analysis suggests that the conjugate hydrolyzed POX through a catalytic mechanism as effective as that of the unconjugated molecules. In summary, we report the first class of dendrimer conjugates which are stable in the plasma and functioning as OP-responsive catalytic scavengers.

 Please wait while we load your content...
Please wait while we load your content...