Synthesis and characterization of protected oligourethanes as crosslinkers of collagen-based scaffolds†
Abstract
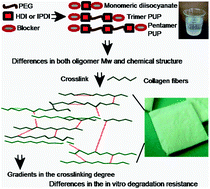
This paper describes the preparation and characterization of water-soluble urethane oligomers bearing protected isocyanate groups. It also points out its ability to crosslink decellularized pericardium, as a model collagen scaffold, and to adjust their structural characteristics. A library of oligourethanes was synthesized by varying the molecular weight (Mw 400, 600, 1000 or 2000 g mol−1) of the poly(ethylene glycol) and the type of aliphatic diisocyanate (isophorone diisocyanate/IPDI or hexamethylene diisocyanate/HDI). 1H and 13C NMR, FTIR and mass spectrometry demonstrated that the crosslinkers are composed of chains with carbamoylsulfonate end groups that have trimeric and pentameric oligourethanes, and monomeric diisocyanate. The degree of crosslinking and hence the in vitro degradation susceptibility of the decellularized pericardium were inversely related to the Mw of the oligourethanes. The toxicity of the extractable products from oligourethane–collagen materials toward fibroblasts and macrophages was found to be lower for the crosslinker derived from IPDI than for those derived from HDI. On the other hand, the resistance to collagenase or oxidative degradation of the bovine pericardium crosslinked with HDI/oligourethane was higher than the one prepared with IPDI/oligourethane. As the Mw of the oligomers regulates the degree of crosslinking while the chemical composition influences the cytocompatibility and biodegradation of decellularized pericardium, these urethane oligomers can be used as safer crosslinkers for other protein-based biomaterials.

 Please wait while we load your content...
Please wait while we load your content...