Physicochemical characterization of segmented polyurethanes prepared with glutamine or ascorbic acid as chain extenders and their hydroxyapatite composites†
Abstract
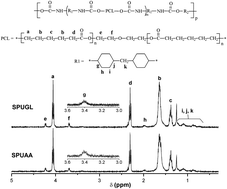
The development of elastomeric, bioresorbable, and biocompatible segmented polyurethanes (SPUs) for use in tissue-engineering applications has attracted considerable interest in recent years because of the existing need for mechanically tunable scaffolds for regeneration of different tissues. In this study segmented polyurethanes were synthesized from poly(ε-caprolactone)diol, 4,4′-methylene bis(cyclohexyl isocyanate) (HMDI) using osteogenic compounds such as ascorbic acid (AA) and L-glutamine (GL) as chain extenders, which are known to play a role in osteoblast proliferation and collagen synthesis. Fourier transform infrared spectroscopy (FTIR) revealed the formation of urethane linkages at 3373, 1729, and 1522 cm−1 (N–H stretching, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching and N–H bending + C–N stretching vibrations, respectively) while urea formation was confirmed by the appearance of a peak at 1632 cm−1. Differential scanning calorimetry, dynamic mechanical analysis, X-ray diffraction and mechanical testing of the polyurethanes showed that these polyurethanes were semi-crystalline polymers (Tg = −25 °C; Tm = 51.4–53.8 °C; 2θ = 21.3° and 23.4°) exhibiting elastomeric behavior (ε > 1000%) only for those prepared by HA incorporation during prepolymer formation. Dense and porous composite matrices of the segmented polyurethanes were prepared by the addition of hydroxyapatite (HA) via either mechanical mixing or in situ polymerization and supercritical fluid processing, respectively. The addition of HA by physical mixing decreased the crystallinity (from 38% to 31%) of the composites prepared with ascorbic acid as the chain extender. Both Tg of the composites and the strain were also lowered to −38 or 36 °C and 27–39% for ascorbic acid and glutamine containing polyurethanes respectively. Composites prepared with ascorbic acid as the chain extender yielded higher Young's modulus and tensile strength than composites prepared with glutamine when HA was incorporated during prepolymer formation. Composites obtained by incorporation of HA by physical mixing revealed a poor dispersion in comparison to composites obtained via HA inclusion during prepolymer formation. In contrast, good dispersion of HA and porosity were achieved at 60 °C, 400 bar and holding times between 0.5 h and 2 h with a downtime between 15 min and 60 min in the CO2 reactor. Biocompatibility studies showed that SPUs containing ascorbic acid allowed the increase of alveolar osteoblast proliferation; hence, they are potentially suitable for bone tissue regeneration.
O stretching and N–H bending + C–N stretching vibrations, respectively) while urea formation was confirmed by the appearance of a peak at 1632 cm−1. Differential scanning calorimetry, dynamic mechanical analysis, X-ray diffraction and mechanical testing of the polyurethanes showed that these polyurethanes were semi-crystalline polymers (Tg = −25 °C; Tm = 51.4–53.8 °C; 2θ = 21.3° and 23.4°) exhibiting elastomeric behavior (ε > 1000%) only for those prepared by HA incorporation during prepolymer formation. Dense and porous composite matrices of the segmented polyurethanes were prepared by the addition of hydroxyapatite (HA) via either mechanical mixing or in situ polymerization and supercritical fluid processing, respectively. The addition of HA by physical mixing decreased the crystallinity (from 38% to 31%) of the composites prepared with ascorbic acid as the chain extender. Both Tg of the composites and the strain were also lowered to −38 or 36 °C and 27–39% for ascorbic acid and glutamine containing polyurethanes respectively. Composites prepared with ascorbic acid as the chain extender yielded higher Young's modulus and tensile strength than composites prepared with glutamine when HA was incorporated during prepolymer formation. Composites obtained by incorporation of HA by physical mixing revealed a poor dispersion in comparison to composites obtained via HA inclusion during prepolymer formation. In contrast, good dispersion of HA and porosity were achieved at 60 °C, 400 bar and holding times between 0.5 h and 2 h with a downtime between 15 min and 60 min in the CO2 reactor. Biocompatibility studies showed that SPUs containing ascorbic acid allowed the increase of alveolar osteoblast proliferation; hence, they are potentially suitable for bone tissue regeneration.

 Please wait while we load your content...
Please wait while we load your content...