Spontaneous interlayer formation in OPVs by additive migration due to additive–metal interactions†
Abstract
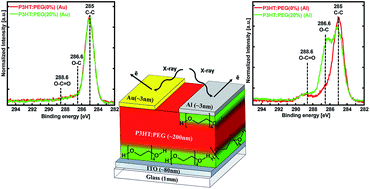
The presence of interlayers between the active layer and the electrode are known to modify the metal work-function and enhance carrier extraction, consequently improving OPV device performance. Spontaneous formation of interlayers by surface-enrichment of suitable additives eliminates separate processing steps and hence is technically advantageous and cost effective. However, surface enrichment is limited to additives with low surface energy. Here we show that additive migration to the organic/electrode interface could be induced by additive–metal interactions, modulated by the interactions between the additive and the underlying substrate. In this study, additive migration induced by metal evaporation is studied by blending P3HT with PEG, an established interlayer material with a surface energy higher than that of P3HT. XPS analysis reveals that, as expected, PEG is not present on the surface of the organic spun film. However, Ca or Al evaporation induces a significant migration of PEG to the organic/metal interface. In contrast, Au evaporation does not induce such migration. The comparison between Al, Ca and Au, metals with significantly different reduction potentials, revealed that the driving force for PEG migration is its chemical interaction with the deposited metal atoms. The extent of PEG migration was also found to depend on the type of underlying substrate, ITO/PEDOT:PSS or ITO. Finally, the PEG interlayer is shown to reduce the Al work function confirming that spontaneous additive migration induced by metal–additive interactions could be harnessed for charge extraction in organic electronic devices.

 Please wait while we load your content...
Please wait while we load your content...