Synthesis of novel platinum complex core as a selective Ag+ sensor and its H-bonded tetrads self-assembled with triarylamine dendrimers for electron/energy transfers†
Abstract
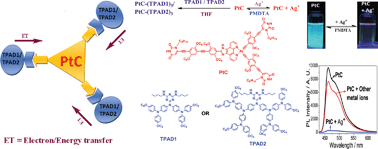
A novel platinum complex PtC with a tri-armed uracil hydrogen-bonded (H-bonded) unit was synthesized via a modular synthetic approach and characterized by 1H, 13C NMR and MALDI-TOF mass spectroscopies. Two H-bonded tetrads, PtC–(TPAD1)3 and PtC–(TPAD2)3, based on a metal core PtC complexed with two generations of triarylamine dendrimers, TPAD1 and TPAD2 (with electron-donating nature), were successfully constructed with improved organic solubility via a classical H-bonded self-assembly approach. Supramolecular H-bonding in solution and solid state was elucidated by 1H NMR titrations, IR spectral studies and time resolved photoluminescence (TRPL) measurements. The electron/energy transfers, as well as the self-assemblies of supramolecular tetrads, were established by UV-Vis and PL titrations and AFM morphological studies. Furthermore, metal complex core PtC showed selective sensitivity towards Ag+ ions through fluorescence turn-off responses without any interference from other common metal ions. The 1 : 1 binding stoichiometry and complexation mechanism between the probe and Ag+ ion was established by 1H NMR titration. Moreover, PL reversibility of PtC + Ag+ could be achieved on addition of PMDTA.

 Please wait while we load your content...
Please wait while we load your content...