CoMn0.2Fe1.8O4 ferrite nanoparticles engineered by sol–gel technology: an expert and versatile catalyst for the reduction of nitroaromatic compounds†
Abstract
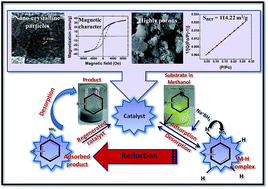
Highly stable and magnetically recoverable CoMn0.2Fe1.8O4 ferrite nanoparticles with a large surface area were successfully engineered using the sol–gel technology and proposed as a heterogeneous catalyst for the reduction of nitroarenes. The morphological and physicochemical properties of the synthesized nanocatalyst were characterized using the powder X-ray diffraction technique, Fourier transform infrared spectroscopy, high resolution transmission electron microscopy, scanning electron microscopy. The nanoparticles were found to be highly porous and spherical with an average diameter of ∼20 nm. The lattice interplanar distance of 0.25, 0.24, 0.30, 0.20 nm determined by high resolution transmission electron microscopy completely agreed with the d-spacing values corresponding to the (3 1 1), (2 2 2), (2 2 0), (4 0 0) planes, respectively, provided by the XRD data. Magnetic characterization was performed using a vibrating sample magnetometer and the value of saturation magnetization was found to be 35.89 emu per g. The specific surface area was calculated using the BET method, which was determined to be 114.22 m2 g−1. Kinetic analysis for the reduction of 2-nitroaniline was performed using UV-visible spectroscopy. The reduction reaction followed pseudo first-order kinetics with respect to the concentration of nitroarene substrate. The pseudo first-order rate constant value was 0.74 min−1 in the presence of 10 mol% of the catalyst. The extent of catalytic activity was explored for various amino, bromo, chloro and methyl derivatives of nitroarenes and almost 100% selectivity and 100% conversions (confirmed by GC-MS) were achieved for all the analogs.

 Please wait while we load your content...
Please wait while we load your content...