One-pot synthesis of microporous carbons highly enriched in nitrogen and their electrochemical performance†
Abstract
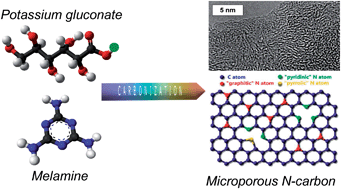
Microporous carbons with large nitrogen contents (6–23 wt%) have been successfully synthesized by the co-carbonization of an alkali organic salt and melamine. In this way, the processes of carbonization, activation and incorporation of nitrogen heteroatoms into the carbon backbone are integrated in only one step. The general applicability of this simple procedure has been proved by using it with a variety of organic salts, including potassium gluconate, and sodium salts of gluconate, citrate and alginate. In particular, the materials produced from potassium gluconate have a narrow micropore size distribution centered at around 0.7–0.8 nm, BET surface areas up to 1040 m2 g−1, and pore volumes of ∼0.3–0.4 cm3 g−1. It was found that the presence of abundant N-groups enhances the electrochemical performance of these materials in 1 M H2SO4. They exhibit high specific capacitances (surface basis) in the 16.6–23 μF cm−2 range and a good electro-oxidation stability as evidenced by the fact that carbon oxidation is shifted to more positive potentials by as much as 500 mV with respect to undoped carbon. In particular, a supercapacitor built with the carbon material synthesized at 850 °C using a melamine/potassium gluconate weight ratio of 2 showed an excellent robustness over a voltage window of 1.2 V in 1 M H2SO4, providing a maximum energy density of 10.2 W h kg−1 (7.9 W h L−1) and a maximum power density of 5.7 kW kg−1 (4.4 kW L−1).

 Please wait while we load your content...
Please wait while we load your content...