Gas sensing properties of Cu2O and its particle size and morphology-dependent gas-detection sensitivity
Abstract
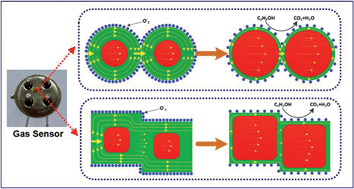
In order to investigate the effect of morphology and size on detection sensitivity of ethanol vapor, nano-scale cubic or spherical Cu2O with similar particle sizes were prepared through reductive solution chemistry routes. Experimental results indicated that both size and morphology of Cu2O particles are playing an important role in gas detection sensitivity, and the mutual competition and mutual dependence relationship coexisted between the two factors. Particle morphology and morphology-dependent particle stacking mode may play a dominant role in a relatively lower gas atmosphere (S50, S260 > C50, C340). As the concentration of ethanol vapor exceeds 200 ppm, the size-dependent effect on gas-sensing gradually appears, and nanoparticles with relatively smaller particle size (C50, S50 > C340, S260) are more advantageous in the gas sensing process. At a much higher ethanol partial pressure, Cu2O nanoparticles with smaller size and cubic morphology exhibit the strongest gas-sensing response. The mechanism of these gas-sensing divergences is suggested to involve in the explanation of effective contact surface, width of charge accumulation layer (CAL) affected by gas concentration, energy bands bending changes and particle contact mode.

 Please wait while we load your content...
Please wait while we load your content...