Identifying pseudocapacitance of Fe2O3 in an ionic liquid and its application in asymmetric supercapacitors†
Abstract
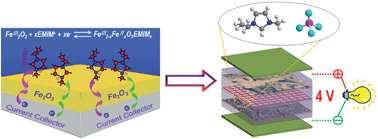
Pseudocapacitance is commonly associated with surface or near-surface reversible redox reactions, as observed with transition metal oxides in alkaline aqueous electrolytes. Here, we demonstrate that pseudocapacitive behavior of Fe2O3 can occur in a 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIMBF4) ionic liquid (IL), and it is closely related to the chemical state variation between Fe3+ and Fe2+ on the surface of a Fe2O3 electrode during the charging/discharging process. By taking advantage of such pseudocapacitance, we prepared a promising electrode material, i.e., graphene nanosheet-supported Fe2O3 nanoparticles (denoted as Fe2O3@GNS), and then built high-performance asymmetric supercapacitors (ASs) using Fe2O3@GNS as the battery-type electrode material, commercial activated carbon (AC)/or activated polyaniline-derived carbon nanorods (denoted as APDC) as the capacitor-type electrode material, and EMIMBF4 IL as the electrolyte. The as-made ASs are able to work reversibly in a full operation voltage region of 0–4 V and exhibit very high energy density. Especially, the AS of Fe2O3@GNS//APDC achieves an extremely high energy density of 177 W h kg−1 and shows a superior combination of high energy and power density (the energy density still remains 62.4 W h kg−1 even at a high power density of 8 kW kg−1).

 Please wait while we load your content...
Please wait while we load your content...