Computational and experimental analysis of Ba0.95La0.05FeO3−δ as a cathode material for solid oxide fuel cells†
Abstract
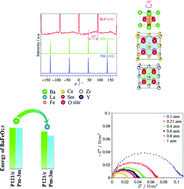
Solid oxide fuel cells (SOFCs) may play a crucial role in solving the energy crisis because they are clean and energy efficient. Finding suitable cathode materials for SOFCs is key to facilitating their widespread use. Besides developing high performance materials, understanding the stability and intrinsic properties of a material is equally important. Herein, Ba0.95La0.05FeO3−δ (BLF) is studied combining molecular simulations and experiments on single crystal thin films. Lattice dynamics simulations are applied to study the stabilization of barium orthoferrate BaFeO3−δ upon doping with La3+. Simulation results reveal the defect energy for substituting one Ba2+ with La3+ in the cubic phase to be lower than that in the monoclinic phase, contributing to its stabilization. Analogous results are also found by doping the Ba site with Sm3+, Gd3+ and Y3+. In addition, the simulation results suggest that the charge compensation mechanism upon doping is filling oxygen vacancies and La3+ tends to trap the mobile oxygen anions. In turn, as the doping level increases the oxygen anion diffusivity decreases, as is also supported by molecular dynamics simulations. In light of this conclusion, single crystal thin films of La3+ slightly doped BaFeO3−δ, BLF, are grown on yttria-stabilized zirconia substrates using pulsed laser deposition. The polarization resistance of the dense film is 0.07 Ω cm2 at 700 °C in an ambient atmosphere, which is comparable to state-of-the-art Co-based materials.

 Please wait while we load your content...
Please wait while we load your content...