Simple small molecule carbon source strategy for synthesis of functional hydrothermal carbon: preparation of highly efficient uranium selective solid phase extractant†
Abstract
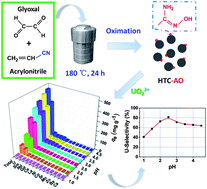
In this paper, simple small molecules, glyoxal and acrylonitrile, are chosen as starting materials to prepare an amidoxime-functionalized hydrothermal carbon-based solid phase extractant (HTC-AO) via a one-step hydrothermal process following a simple oximation. The resulting HTC-AO exhibits the anticipated properties, i.e., low porosity (0.01 cm3 g−1) and intraparticle diffusion coefficient (kint = 0.042 mmol g−1 min−0.5), high content of amidoxime groups (1.66 mmol g−1) and minimal undesired functional groups (typically carboxylic group: 0.07 mmol g−1; phenolic group: 0.38 mmol g−1; lactonic group: 0.01 mmol g−1). Moreover, the results of irradiation experiments under γ-ray dosages between 1 and 100 kGy indicate that HTC-AO has good radiation stability. The sorption behavior of U(VI) onto HTC-AO is investigated in detail using batch sorption experiments. A saturation U(VI) sorption capacity over that of all the uranium sorbents reported previously is found to be 1021.6 mg g−1 at pH 4.5 in single uranium solution, and a so far unreported highest uranium selectivity of 81.6% with a sorption capacity of 268.9 mg g−1 is observed at pH 2.5 in multi-ion solution. The significant outcomes in this work confirms that the “simple small molecule carbon source” strategy is practical and efficient, and may have the potential for the preparation of other types of functional materials such as highly specific catalysts, drug targeting carriers and others.

 Please wait while we load your content...
Please wait while we load your content...