Dichotomous adsorption behaviour of dyes on an amino-functionalised metal–organic framework, amino-MIL-101(Al)†
Abstract
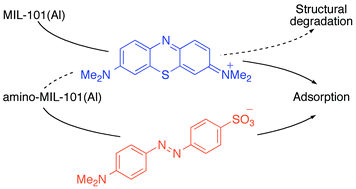
An amino-functionalised metal–organic framework (MOF), aluminium aminoterephthalate (amino-MIL-101(Al)), has been applied to the adsorptive removal of dyes (cationic methylene blue, MB, and anionic methyl orange, MO) from aqueous solutions in order to examine the effect of the amino group on sorption behaviour. Adsorption isotherms and thermodynamic studies indicated the spontaneous adsorption of MB with a maximum adsorption capacity at 30 °C (762 ± 12 mg gMOF−1) higher than those observed for MB on other MOFs and most other materials. In contrast, lower adsorption capacities were observed in the adsorption of the same dye on the analogous non-amino-functionalised framework (MIL-101(Al), 195 mg g−1) and in the adsorption of MO by amino-MIL-101(Al) (188 ± 9 mg g−1), suggesting that an electrostatic interaction between the amino groups of the MOF and the cationic dye MB may have contributed to the high adsorption capacity. The adsorptions of both dyes on amino-MIL-101(Al) were spontaneous, endothermic, and entropy-driven, as is common for dye adsorptions. However, the ΔS value obtained for the adsorption of MB (346 J mol−1 K−1) was extreme. Further analysis demonstrated that after exposure to MB, the ordered amino-MIL-101(Al) structure was absent, ∼30% of the Al3+ was lost to solution, and significant changes occurred in the X-ray photoelectron spectrum of the MOF. On the other hand, the MOF structure was intact following the adsorption of MO. Several groups have exploited electrostatic interactions to improve dye adsorption; however, these proved excessive in the case of MB (but not MO) adsorption on amino-MIL-101(Al).

 Please wait while we load your content...
Please wait while we load your content...