Phosphorus(v) tetraazaporphyrins: porphyrinoids showing an exceptionally strong CT band between the Soret and Q bands†
Abstract
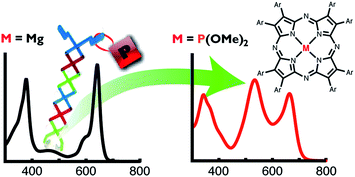
A series of tetraazaporphyrin phosphorus(V) complexes (PTAPs) has been synthesized and characterized practically for the first time, and these complexes are found to exhibit unique absorption properties not observed for normal porphyrinoids. The absorption spectrum of [Ar8TAPP(OMe)2][ClO4] shows a broad, intense band between the Soret and Q bands at around 530 nm, differing from the typical metalloTAPs and phthalocyanine phosphorus(V) complexes (PPcs). Both electron-donating and -withdrawing substituents affect the complete absorption envelope of the PTAPs, even though the substituents lie outside the TAP π-conjugation system. Moreover, the trends across the entire absorption range correlate well with the Hammett constants corresponding to the substituents. Magnetic circular dichroism (MCD), cyclic voltammetry (CV) and theoretical calculations using density functional theory (DFT) were performed to interpret the unique absorption properties of the PTAPs. The interaction between the aryl substituents and the TAP macrocyclic core could be estimated by carefully taking into account the results of spectroscopies and frontier molecular orbitals. The effect of the phosphorus(V) ion in the TAP was found to be stronger than that in the phthalocyanine (Pc). In the TAP, the phosphorus(V) ion intensifies a charge-transfer (CT) band from the peripheral aryl moieties to the TAP core. Band deconvolution analysis of the absorption and MCD spectra also clarified the effect of the phosphorus(V) ion. The combination of a main-group element (phosphorus) and a macrocyclic core (TAP) appears useful for developing new organic materials which absorb over the complete UV-visible region (300–700 nm) as a single, discrete compound.

 Please wait while we load your content...
Please wait while we load your content...