The role of 5f-orbital participation in unexpected inversion of the σ-bond metathesis reactivity trend of triamidoamine thorium(iv) and uranium(iv) alkyls†
Abstract
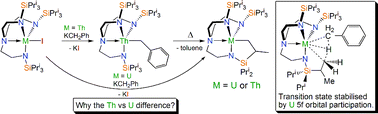
We report on the role of 5f-orbital participation in the unexpected inversion of the σ-bond metathesis reactivity trend of triamidoamine thorium(IV) and uranium(IV) alkyls. Reaction of KCH2Ph with [U(TrenTIPS)(I)] [2a, TrenTIPS = N(CH2CH2NSiPri3)33−] gave the cyclometallate [U{N(CH2CH2NSiPri3)2(CH2CH2NSiPri2C[H]MeCH2)}] (3a) with the intermediate benzyl complex not observable. In contrast, when [Th(TrenTIPS)(I)] (2b) was treated with KCH2Ph, [Th(TrenTIPS)(CH2Ph)] (4) was isolated; which is notable as Tren N-silylalkyl metal alkyls tend to spontaneously cyclometallate. Thermolysis of 4 results in the extrusion of toluene and formation of the cyclometallate [Th{N(CH2CH2NSiPri3)2(CH2CH2NSiPri2C[H]MeCH2)}] (3b). This reactivity is the reverse of what would be predicted. Since the bonding of thorium is mainly electrostatic it would be predicted to undergo facile cyclometallation, whereas the more covalent uranium system might be expected to form an isolable benzyl intermediate. The thermolysis of 4 follows well-defined first order kinetics with an activation energy of 22.3 ± 0.1 kcal mol−1, and Eyring analyses yields ΔH‡ = 21.7 ± 3.6 kcal mol−1 and ΔS‡ = −10.5 ± 3.1 cal K−1 mol−1, which is consistent with a σ-bond metathesis reaction. Computational examination of the reaction profile shows that the inversion of the reactivity trend can be attributed to the greater f-orbital participation of the bonding for uranium facilitating the σ-bond metathesis transition state whereas for thorium the transition state is more ionic resulting in an isolable benzyl complex. The activation barriers are computed to be 19.0 and 22.2 kcal mol−1 for the uranium and thorium cases, respectively, and the latter agrees excellently with the experimental value. Reductive decomposition of “[U(TrenTIPS)(CH2Ph)]” to [U(TrenTIPS)] and bibenzyl followed by cyclometallation to give 3a with elimination of dihydrogen was found to be endergonic by 4 kcal mol−1 which rules out a redox-based cyclometallation route for uranium.

 Please wait while we load your content...
Please wait while we load your content...