G-quadruplex-binding ligand-induced DNA synapsis inside a DNA origami frame†
Abstract
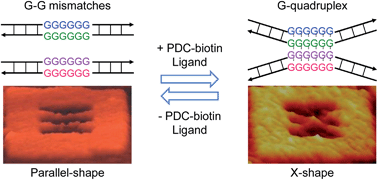
Among the approaches for DNA-based drug targeting, G-quadruplex-binding ligands are of particular interest because of the high abundance of G-rich sequences in regions such as human chromosomal telomeres and promoters of several proto-oncogenes. A number of quadruplex-ligands have been reported, but their functions at single-molecule level have not been explored using direct and real-time methods. Here, we report on the direct observation of the formation of a four-stranded G-quadruplex induced by bisquinolinium pyridine dicarboxamide with a linker containing biotin at one end. We fabricated a DNA origami frame with incorporated duplex DNAs that contained 3–6 G–G mismatches in the middle. In the absence of ligand, the duplex DNAs of interest had no interaction, as visualized by their parallel-shape in high-speed atomic force microscopy (HS-AFM) images. Presence of ligand induced the formation of G-quadruplex structure, which was characterized by an X-shape. Addition of streptavidin to the ligand-induced quadruplex caused the protein to localize in the middle of the X-shape, indicating that the ligand is bound to the quadruplex. A sequence of real-time images of the ligand-induced formation of a quadruplex and its reverse conformational switching by removing the ligand was captured by HS-AFM. Unprecedented intermediate-like states were recorded in our real-time analysis.

 Please wait while we load your content...
Please wait while we load your content...