Aerobic oxidation of aldehydes: selectivity improvement using sequential pulse experimentation in continuous flow microreactor
Abstract
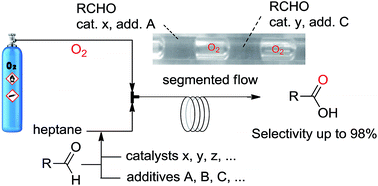
The aerobic oxidation of aldehyde was investigated using a continuous flow microreactor under 5 bar of oxygen at room temperature. High-throughput screening of experimental conditions resulted in the development of an improved protocol. The synergistic use of a large range of salts and Mn(II) catalyst was found to be a very efficient catalytic system for selective aldehyde oxidation. Indeed for short residence time (i.e. 6 min.), a quantitative conversion of 2-ethylhexanal was obtained with a selectivity toward carboxylic acid of 98%.

 Please wait while we load your content...
Please wait while we load your content...