Au–Cu–Pt ternary catalyst fabricated by electrodeposition and galvanic replacement with superior methanol electrooxidation activity
Abstract
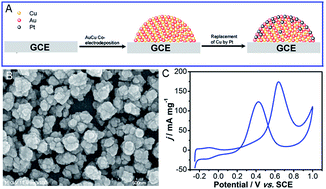
A series of Au–Cu–Pt ternary catalysts were fabricated on glassy carbon electrode (GCE) by a two-step method. Au–Cu nanoparticles were formed by initial electrodeposition of Au–Cu layers onto GCE and then followed by the partial replacement of Cu by Pt. The morphology and composition of Au–Cu–Pt catalysts were characterized by scanning electron microscopy (SEM), energy dispersive X-ray analysis (EDX), EDX element mapping and X-ray photoelectron spectroscopy (XPS). It was found that the alloying micro-structures existed in the catalysts among Au, Cu and/or the partially replaced Pt. Moreover, electrochemical measurements revealed that despite low loading of Pt, the Au–Cu–Pt/GCE-10 catalyst presented superior electrocatalytic activity and stability to that of the other comparative electrodes toward methanol electrooxidation (MEO). It indicated that this two-step method can efficiently decrease the amount of Pt loading in the catalyst. These findings also suggested that the prepared Au–Cu–Pt catalyst has a great potential for use in the direct methanol fuel cell (DMFC).

 Please wait while we load your content...
Please wait while we load your content...