Facile and large-scale synthesis of poly(m-phenylenediamine) nanobelts with high surface area and superior dye adsorption ability†
Abstract
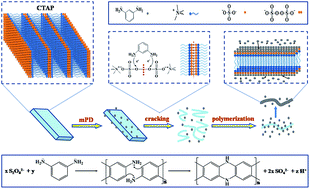
Uniform poly(m-phenylenediamine) (PmPD) nanobelts have been synthesized through chemical oxidative polymerization of m-phenylenediamine by using white cetyl trimethyl ammonium persulfate (CTAP) powders as oxidants. Results from the Brunauer–Emmett–Teller (BET) method indicated that the surface area of PmPD reached as high as 284.5 m2 g−1. The influence of high surface area on Orange G adsorption was examined. The Orange G adsorbance of PmPD can reach 469.5 mg g−1. The adsorption process can be better described by the Langmuir and pseudo-second-order kinetic model. The acid doping of PmPD nanobelts could produce better adsorption performance for Orange G The superior performance of PmPD nanobelts makes them hopeful functional materials for dye removal.

 Please wait while we load your content...
Please wait while we load your content...