Catalytic oxidation of acetone over CuCeOx nanofibers prepared by an electrospinning method
Abstract
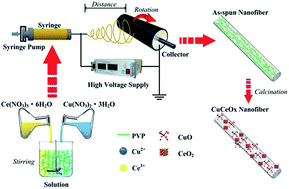
A series of CuCeOx nanofiber catalysts with different Cu/(Cu + Ce) molar ratios were synthesized by an electrospinning method. The catalysts were evaluated for acetone oxidation at different temperatures (100–280 °C) with a GHSV of 79 000 ml g−1 h−1. The results showed that nanofiber catalysts possessed better catalytic performance than catalysts prepared by urea-nitrate combustion and sol–gel methods. An appropriate Cu/(Cu + Ce) molar ratio could greatly improve the activity of the nanofiber catalysts, and a significant improvement of the activity was obtained on the Cu0.50Ce0.50Ox nanofiber catalyst (∼100% acetone conversion at 270 °C). Characteristic analysis of prepared catalysts suggested (1) a special nanofibrous morphology with large specific surface area; (2) abundant oxygen vacancies and (3) cerium ions with unusual oxidation states were the main factors that would affect catalytic activity of CuCeOx nanofibers catalysts.

 Please wait while we load your content...
Please wait while we load your content...