Fast photocatalytic degradation of congo red using CoO-doped β-Ga2O3 nanostructures†
Abstract
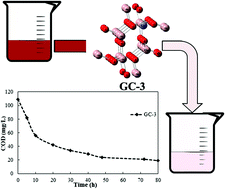
In this investigation, the influence of cobalt(II) oxide on the photocatalytic activity of Ga2O3 rod-like nanostructures in the presence of 30 ppm congo red is studied. Samples of Ga2O3 doped with 1, 2, 3 and 5 wt% CoO were synthesized by employing a facile co-precipitation method using aqueous solutions of Co(II) chloride and gallium nitrate and CTAB as a structure directing agent and characterized by X-ray diffraction, energy-dispersive X-ray, scanning electron microscopy, photoluminescence, BET surface area, UV-vis diffuse reflectance spectroscopy and point of zero charge techniques. The samples containing up to 2 wt% of CoO calcined at 900 °C revealed that only a monoclinic structure of Ga2O3 is formed. Despite equal phase of β-Ga2O3, two other phases of CoGa2O4 and α-Ga2O3 were observed in X-ray diffraction patterns of samples containing 3 and 5 wt% of CoO, respectively. UV-vis diffuse reflectance spectroscopy showed a significant reduction in band gap upon increasing their CoO content, indicating incorporation of CoO in the gallia lattice. The sample containing 3 wt% CoO–Ga2O3 can remove 30 ppm of congo red readily within 75 minutes and it exhibited a high photo-degradation activity of about 82.5% after 80 h of irradiation. These results suggest that CoO–gallia/UV photocatalysis may be envisaged as a method for treatment of diluted colored waste water not only for decolorization, but also for detoxification in textile industries. The photocatalytic degradation of the dye in solution obeys first order reaction kinetics.

 Please wait while we load your content...
Please wait while we load your content...