Chemically cross-linked microtubule assembly shows enhanced dynamic motions on kinesins†
Abstract
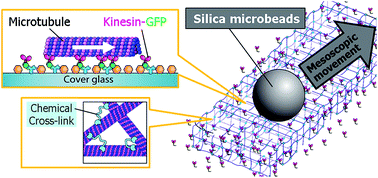
Biomolecular motors have cooperative functions that facilitate various important cellular functions such as signaling and deformation. However, the multiple-molecular behavior is poorly understood when compared with the well-studied behavior of single molecules. Kinesin and microtubules (MTs) are a combination of motor and rail proteins that have been reported to show cooperativity in moving patterns. Here, we report that cross-linking of isotropic MTs enhances the movement of the local network temporally, which can elicit the effective conveyance of 5 μm diameter silica microbeads. The average velocity of the beads reached 143 nm s−1 for over 15 min, which is 2.7 times higher than the beads on kinesin-driven MTs without cross-links. Although the velocity of the beads fluctuated over the time course, the maximum velocity calculated from 5 s displacement reached 1.2 μm s−1, which was ca. 3-fold higher than the intrinsic driving velocity of kinesins, i.e., the sliding velocity of the MTs without cross-links. This technique provides such an enhanced movement by simply cross-linking the isotropic MTs, but without specific control or alignment over network polarity. The enhancement in the local motion of the MTs is discussed in terms of the “mechanochemical” effect coupled between dynamically stored elastic energies in the MT network and the collective motion of MTs driven by an ATP-fueled kinesin motor.

 Please wait while we load your content...
Please wait while we load your content...